Publications
Group Highlights
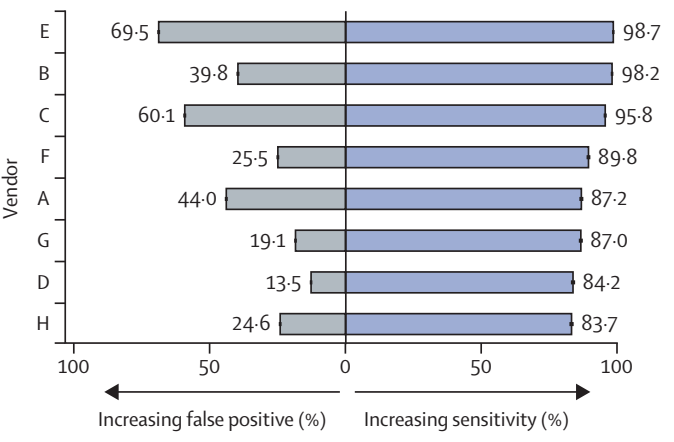
The global prevalence of diabetes is rising, alongside costs and workload associated with screening for diabetic eye disease (diabetic retinopathy). Automated retinal image analysis systems (ARIAS) could replace primary human grading of images for diabetic retinopathy. We evaluated multiple ARIAS in a real-life screening programme. Eight of 25 invited and potentially eligible CE-marked systems for diabetic retinopathy detection from retinal images agreed to participate. From 202,886 screening encounters at the North East London Diabetic Eye Screening Programme (between Jan 1, 2021, and Dec 31, 2022) we curated a database of 1·2 million images and sociodemographic and grading data. Images were manually graded by up to three graders according to a standard national protocol. ARIAS performance overall and by subgroups of age, sex, ethnicity, and index of multiple deprivation (IMD) were assessed against the reference standard.
A. R. Rudnicka et al.
Lancet Digit. Health, vol. 7, no. 11, 2025.
Retinal imaging offers a non-invasive means to assess the circulatory system, with morphological features of retinal vessels serving as biomarkers for systemic disease. QUARTZ (QUantitative Analysis of Retinal vessel Topology and siZe) is a fully automated artificial intelligence-enabled retinal vasculometry system designed to process large-scale retinal image datasets to obtain quantitative measures of vessel morphology for use in epidemiological studies. Previously reliant on traditional image processing and machine learning, QUARTZ has now transitioned to a deep learning pipeline.
R. A. Welikala et al.
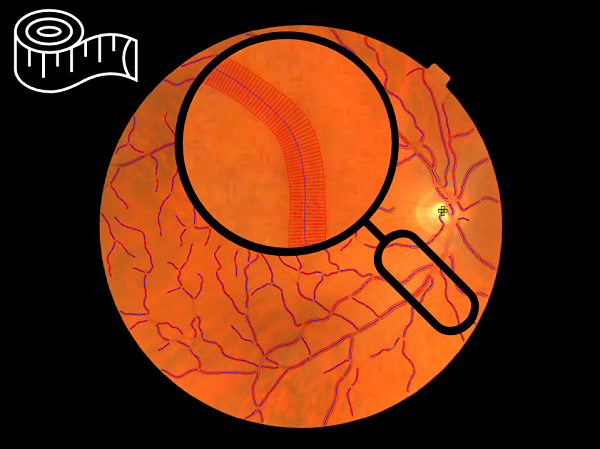
We examine whether inclusion of artificial intelligence (AI)-enabled retinal vasculometry (RV) improves existing risk algorithms for incident stroke, myocardial infarction (MI) and circulatory mortality. AI-enabled retinal vessel image analysis processed images from 88 052 UK Biobank (UKB) participants (aged 40–69 years at image capture) and 7411 European Prospective Investigation into Cancer (EPIC)-Norfolk participants (aged 48–92). Retinal arteriolar and venular width, tortuosity and area were extracted. Prediction models were developed in UKB using multivariable Cox proportional hazards regression for circulatory mortality, incident stroke and MI, and externally validated in EPIC-Norfolk.
A. R. Rudnicka et al.
British Journal of Ophthalmology, vol. 106, no. 12, pp. 1722–1729, 2022.
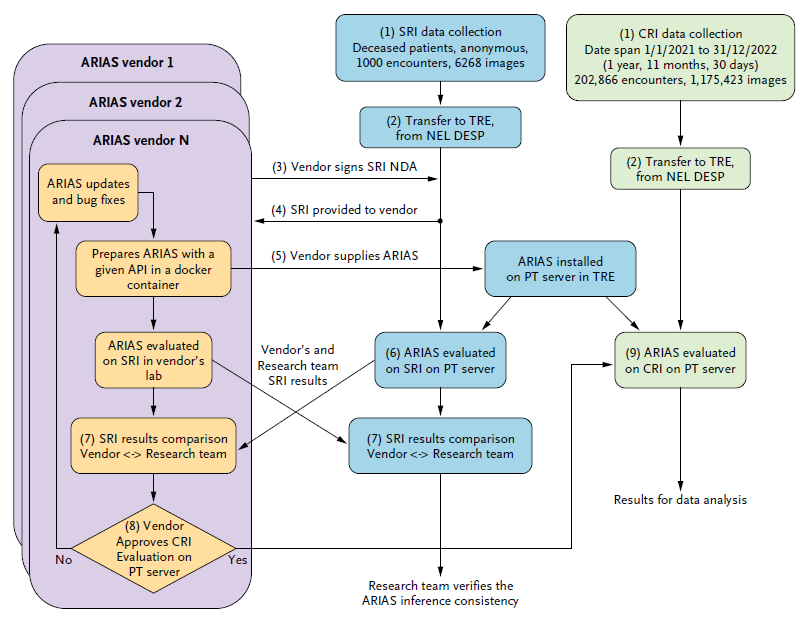
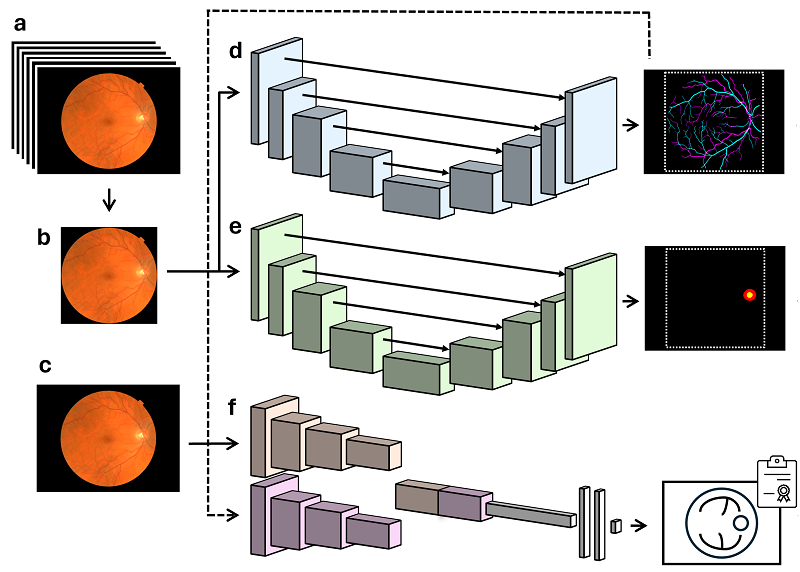
The deployment of algorithms in health care screening programs has been hindered by a lack of agreed-upon methodology to evaluate trustworthiness and equity. We outline transferable methodology for independent evaluation of algorithms using a routine, high-volume, multiethnic national diabetic eye screening program as an exemplar. Automated retinal image analysis systems (ARIAS), including artificial intelligence (AI), for detection of diabetic retinopathy (DR) could substantially increase image-grading capacity. We report technical and operational considerations relevant to implementation and evaluation in large-scale population screening.
J. Fajtl et al.
NEJM AI, vol. 1, no. 9, p. AIoa2400353, 2024.
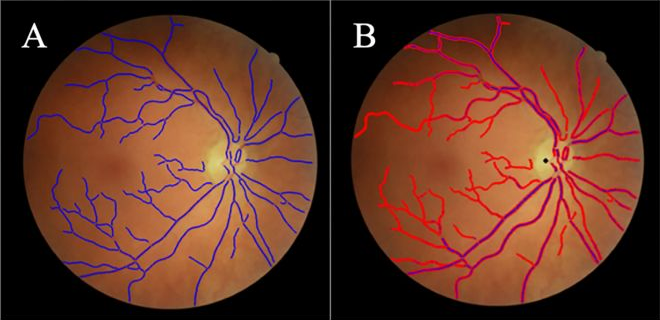
To evaluate the test performance of the QUARTZ (QUantitative Analysis of Retinal vessel Topology and siZe) software in detecting retinal features from retinal images captured by health care professionals in a Danish high street optician chain, compared with test performance from other large population studies (i.e., UK Biobank) where retinal images were captured by non-experts.
J. Freiberg et al.
PLoS One, vol. 18, no. 8, p. e0290278, 2023.
Selected Publications
-
A. R. Rudnicka et al., ‘Automated retinal image analysis systems to triage for grading of diabetic retinopathy: a large-scale, open-label, national screening programme in England’, Lancet Digit. Health, vol. 7, no. 11, 2025.
-
A. Rudnicka et al., ‘Is Artificial intelligence (AI) enabled retinal vasculometry key to the identification age, sex and race from retinal images?’, Invest Ophthalmol Vis Sci, vol. 66, no. 8, p. 3875, 2025.
-
A. O’Neil et al., ‘Retinal Vessel Traits and Age‐Related Eye Disease in the Canadian Longitudinal Study on Aging’, Clin Exp Ophthalmol, 2025.
-
A. O’Neil et al., ‘Factors Associated With Retinal Vessel Traits in the Canadian Longitudinal Study on Aging’, Invest Ophthalmol Vis Sci, vol. 66, no. 3, p. 13, 2025.
-
J. Freiberg et al., ‘Longitudinal associations of retinal vessel morphology with intraocular pressure and blood pressure at follow‐up visit—Findings from a Danish eye and vision cohort, Project FOREVER’, Acta Ophthalmol, vol. 103, no. 1, pp. 33–42, 2025.
-
R. Shakespeare et al., ‘Retinal vasculometry associations with cognition status in UK Biobank’, Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring, vol. 17, no. 1, p. e270087, 2025.
-
R. A. Welikala et al., ‘Artificial intelligence-enabled retinal vasculometry at scale utilizing the UK Biobank, CLSA, and NEL DESP datasets’, in 2024 IEEE EMBS International Conference on Biomedical and Health Informatics (BHI), IEEE, 2024, pp. 1–8.
-
J. Fajtl et al., ‘Trustworthy evaluation of clinical AI for analysis of medical images in diverse populations’, NEJM AI, vol. 1, no. 9, p. AIoa2400353, 2024.
-
J. Freiberg et al., ‘Automated analysis of vessel morphometry in retinal images from a Danish high street optician setting’, PLoS One, vol. 18, no. 8, p. e0290278, 2023.
-
X. Jiang et al., ‘GWAS on retinal vasculometry phenotypes’, PLoS Genet, vol. 19, no. 2, p. e1010583, 2023.
-
A. R. Rudnicka et al., ‘Artificial intelligence-enabled retinal vasculometry for prediction of circulatory mortality, myocardial infarction and stroke’, British Journal of Ophthalmology, vol. 106, no. 12, pp. 1722–1729, 2022.
-
R. J. Tapp et al., ‘Retinal microvascular associations with cardiometabolic risk factors differ by diabetes status: results from the UK Biobank’, Diabetologia, vol. 65, no. 10, pp. 1652–1663, 2022.
-
A. R. Rudnicka et al., ‘Retinal vasculometry associations with glaucoma: Findings from the european prospective investigation of cancer–norfolk eye study’, Am J Ophthalmol, vol. 220, pp. 140–151, 2020.
-
R. J. Tapp et al., ‘Retinal vascular tortuosity and diameter associations with adiposity and components of body composition’, Obesity, vol. 28, no. 9, pp. 1750–1760, 2020.
-
M. Yates et al., ‘Retinal vasculometric characteristics and their associations with polymyalgia rheumatica and giant cell arteritis in a prospective cohort: EPIC-Norfolk Eye Study’, Ann Rheum Dis, vol. 79, no. 4, 2020.
-
R. J. Tapp et al., ‘Associations of retinal microvascular diameters and tortuosity with blood pressure and arterial stiffness: United Kingdom Biobank’, Hypertension, vol. 74, no. 6, pp. 1383–1390, 2019.
-
C. G. Owen et al., ‘Retinal Vasculometry Associations with Cardiometabolic Risk Factors in the European Prospective Investigation of Cancer—Norfolk Study’, Ophthalmology, vol. 126, no. 1, 2019.
-
R. A. Welikala et al., ‘Automated quantification of retinal vessel morphometry in the UK biobank cohort’, in 2017 Seventh International Conference on Image Processing Theory, Tools and Applications (IPTA), IEEE, 2017, pp. 1–6.
-
R. A. Welikala et al., ‘Automated arteriole and venule classification using deep learning for retinal images from the UK Biobank cohort’, Comput Biol Med, vol. 90, 2017.
-
M. M. Habib et al., ‘Incorporating spatial information for microaneurysm detection in retinal images’, Advances in Science, Technology and Engineering Systems Journal, vol. 2, no. 3, pp. 642–649, 2017.
-
M. M. Habib, R. A. Welikala, A. Hoppe, C. G. Owen, A. R. Rudnicka, and S. A. Barman, ‘Detection of microaneurysms in retinal images using an ensemble classifier’, Inform Med Unlocked, vol. 9, 2017.
-
R. A. Welikala et al., ‘Automated retinal image quality assessment on the UK Biobank dataset for epidemiological studies’, Comput Biol Med, vol. 71, pp. 67–76, 2016.
-
M. Yates, ‘Retinal vessel morphometry associations with polymyalgia rheumatica; findings from the European Prospective Investigation of Cancer (EPIC) in Norfolk’, American College of Rheumatology, 2016.
-
M. M. Fraz, R. A. Welikala, A. R. Rudnicka, C. G. Owen, D. P. Strachan, and S. A. Barman, ‘QUARTZ: Quantitative analysis of retinal vessel topology and size - An automated system for quantification of retinal vessels morphology’, Expert Syst Appl, vol. 42, no. 20, pp. 7221–7234, 2015.
-
R. A. Welikala et al., ‘Automated retinal vessel recognition and measurements on large datasets’, in Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBS, 2015, pp. 5239–5242.
-
R. A. Welikala et al., ‘Genetic algorithm based feature selection combined with dual classification for the automated detection of proliferative diabetic retinopathy’, Computerized Medical Imaging and Graphics, vol. 43, pp. 64–77, 2015.
-
R. A. Welikala et al., ‘Automated detection of proliferative diabetic retinopathy using a modified line operator and dual classification’, Comput Methods Programs Biomed, vol. 114, no. 3, pp. 247–261, 2014.
-
M. M. Fraz, P. H. Whincup, and S. A. Barman, ‘Quantification of blood vessel calibre in retinal images of multi-ethnic school children using a model based approach’, Computerized Medical Imaging and Graphics, vol. 37, no. 1, pp. 48–60, 2013.
-
M. M. Fraz et al., ‘An ensemble classification-based approach applied to retinal blood vessel segmentation’, IEEE Trans Biomed Eng, vol. 59, no. 9, pp. 2538–2548, 2012.
-
C. G. Owen et al., ‘Retinal arteriolar tortuosity and cardiovascular risk factors in a multi-ethnic population study of 10-year-old children; the Child Heart and Health Study in England (CHASE)’, Arterioscler Thromb Vasc Biol, vol. 31, no. 8, pp. 1933–1938, 2011.
-
C. G. Owen et al., ‘Measuring retinal vessel tortuosity in 10-year-old children: validation of the computer-assisted image analysis of the retina (CAIAR) program’, Invest Ophthalmol Vis Sci, vol. 50, no. 5, pp. 2004–2010, 2009.